Class 8 Science Chapter 9 The Amazing World of Solutes, Solvents, and Solutions Question Answer
Science Class 8 Chapter 9 Question Answer The Amazing World of Solutes, Solvents, and Solutions
The Amazing World of Solutes, Solvents, and Solutions Class 8 Question Answer (InText)
Question 1.
We know air is a mixture. Would a mixture of gases also be considered a solution? (Page 135)
Answer:
Yes, if it is a uniform mixture.
Question 2.
What will happen if we keep adding more salt to a given amount of water? (Page 136)
Answer:
It will dissolve to its maximum solubility and then settle down.
Question 3.
Do gases also dissolve in water? (Page 139)
Answer:
Oxygen dissolves in water to a low extent only.
![]()
Question 4.
Why are measuring cylinders always designed narrow and tall instead of wider and shorter like a beaker? (Page 144)
Answer:
Narrow and tall cylinders give a better measurement of volume.
Question 5.
I wonder how the level of a coloured liquid is measured? (Page 145)
Answer:
In case of coloured liquid, the mark on the measuring cylinder should coincide with the top of the meniscus.
NCERT Class 8 Science Chapter 9 Question Answer The Amazing World of Solutes, Solvents, and Solutions (Exercise)
Keep the Curiosity Alive (Pages 149-151)
Question 1.
State whether the statements given below are True [T] or False [F]. Correct the false statements.
(i) Oxygen gas is more soluble in hot water than in cold water.
(ii) A mixture of sand and water is a solution.
(iii) The amount of space occupied by any object is called its mass.
(iv) An unsaturated solution has more solute dissolved than a saturated solution.
(v) The mixture of different gases in the atmosphere is also a solution.
Answer:
(i) False: Oxygen is more soluble in cold water.
(ii) False: A Mixture of sand and water is not a solution. Sand does not dissolve in water but settles down.
(iii) False: The amount of space occupied by any object is called its volume.
(iv) False: A saturated solution has more solute dissolved than an unsaturated solution.
(v) True.
Question 2.
Fill in the blanks:
(i) The volume of a solid can be measured by the method of displacement, where the solid is ____________ in water and the ____________ in water level is measured.
(ii) The maximum amount of ____________ dissolved in ____________ at a particular temperature is called solubility at that temperature.
(iii) Generally, the density ____________ with increase in temperature.
(iv) The solution in which glucose has completely dissolved in water, and no more glucose can dissolve at a given temperature, is called a ____________ solution of glucose.
Answer:
(i) placed; rise
(ii) solute, solvent
(iii) decrease
(iv) saturated
Question 3.
You pour oil into a glass containing some water. The oil floats on top. What does this tell you?
(i) Oil is denser than water.
(ii) Water is denser than oil.
(iii) Oil and water have the same density.
(iv) Oil dissolves in water.
Answer:
(ii) Water is denser than oil.
![]()
Question 4.
A stone sculpture weighs 225 g and has a volume of 90 cm³. Calculate its density and predict whether it will float or sink in water.
Answer:
Density of stone = \(\frac {225}{90}\) = 2.5 g/cm³
It will sink in water.
Question 5.
Which one of the following is the most appropriate statement, and why are the other statements not appropriate?
(i) A saturated solution can still dissolve more solute at a given temperature.
(ii) An unsaturated solution has dissolved the maximum amount of solute possible at a given temperature.
(iii) No more solute can be dissolved into the saturated solution at that temperature.
(iv) A saturated solution forms only at high temperatures.
Answer:
Statement (iii) is most appropriate.
(i) It is not appropriate as a saturated solution cannot dissolve more solute at a given temperature.
(ii) An unsaturated solution can have more solute dissolved at a given temperature.
(iii) Correct.
(iv) A Saturated solution can be formed at all temperatures.
Question 6.
You have a bottle with a volume of 2 litres. You pour 500 mL of water into it. How much more water can the bottle hold?
Answer:
The bottle of 2 litres capacity can hold 1500 mL more of water besides 500 mL.
Question 7.
An object has a mass of 400 g and a volume of 40 cm³. What is its density?
Answer:
Density = \(\frac {mass}{volume}\)
= \(\frac {400}{40}\)
= 10 g/cm³
Question 8.
Analyse Figures (a) and (b). Why does the unpeeled orange float, while the peeled one sinks? Explain.
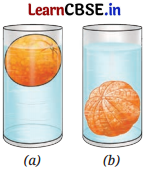
Answer:
An unpeeled orange displaces more water, and so it floats. Peeled orange displaces less water than its weight, so it sinks.
Question 9.
Object A has a mass of 200 g and a volume of 40 cm³. Object B has a mass of 240 g and a volume of 60 cm³. Which object is denser?
Answer:
Density of object A = \(\frac {200}{40}\) = 5 g/cm³
Density of object B = \(\frac {240}{60}\) = 4 g/cm³
Conclusion: Object A is denser, having more density than B.
Question 10.
Reema has a piece of modeling clay that weighs 120 g. She first moulds it into a compact cube that has a volume of 60 cm³. Later, she flattens it into a thin sheet. Predict what will happen to its density.
Answer:
The density of flattened clay will decrease as it displaces more liquid.
![]()
Question 11.
A block of iron has a mass of 600 g and a density of 7.9 g/cm³. What is its volume?
Answer:
We know density = \(\frac {mass}{volume}\)
∴ Volume = \(\frac {mass}{density}\)
= \(\frac{600 \mathrm{~g}}{7.9 \mathrm{~g} / \mathrm{cm}^3}\)
= 75.94 cm³
Question 12.
You are provided with an experimental setup as shown in Figures (a) and (b). On keeping the test tube (Figure b) in a beaker containing hot water (~ 70°C), the water level in the glass tube rises. How does it affect the density?
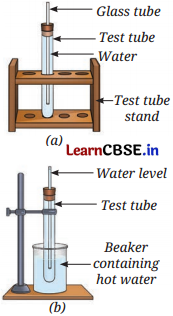
Answer:
The density of water in setup (b) will decrease.
Class 8 The Amazing World of Solutes, Solvents, and Solutions Question Answer (Activities)
Activity 9.1: Let us Investigate (Page 136)
Take a clean glass tumbler and fill it half with water.
Add one spoon of salt into it and stir well till it dissolves completely.

Gradually add a spoonful of salt into the glass tumbler and stir.
Observe how many spoons of salt you can add before it stops dissolving completely.
Record your observations in the Table.
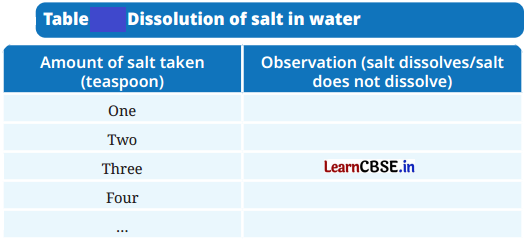
Answer:
Quite a few spoons of salt could be dissolved before some of it remained undissolved.
Water has a high capacity to dissolve salts.
Activity 9.2: Let us Experiment (Demonstration Activity) (Page 137)
Take about 50 mL of water in a glass beaker and measure its temperature using a laboratory thermometer, say 20°C. Add a spoonful of baking soda (sodium hydrogen carbonate) to the water and stir until it dissolves. Continue adding small amounts of baking soda while stirring, till some solid baking soda is left undissolved at the bottom of the beaker. Now, heat the contents to 50°C while stirring (figure).
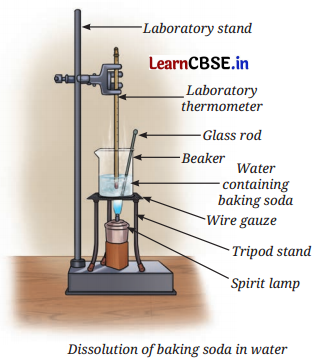
What happens to the undissolved baking soda?
You will observe that it has dissolved.
Continue adding more baking soda while stirring at this temperature until some solid baking soda remains undissolved.
Again, heat the contents further to 70°C while continuing to stir. What do you observe?
The undissolved baking soda dissolves.
What do you infer from this experiment?
Answer:
Water at 70°C dissolves more of the baking soda than at 50°C. It is inferred that for most of the substances, the solubility increases with temperature. It can also be concluded that a saturated solution becomes an unsaturated solution at a higher temperature.
![]()
Activity 9.3: Let us Measure (Page 142)
Switch ON the digital weighing balance.
Note the reading displayed on the balance, which gives the mass of the stone, say 16.400 g.
Answer:
The difference measured between the weight of the watch glass and the watch glass with the object gives the weight/mass of the object. In case of liquids, a watch glass is replaced with a measuring bottle.
Activity 9.4: Let us Observe and Calculate (Page 143)
What is the maximum volume it can measure?
Answer:
The maximum volume that can be measured in a cylinder depends on its size.
How much is the volume difference indicated between the two bigger marks (for example, between 10 mL and 20 mL)?
Answer:
The volume difference between two bigger marks (say 10 mL and 20 mL) is 10 mL.
How many smaller divisions are there between the two bigger marks?
Answer:
There are 10 smaller divisions between the two bigger marks.
How much volume does one small division indicate?
Answer:
One small division indicates 0.1 mL.
The smallest volume that the measuring cylinder can read is 1 mL.
Activity 9.5: Let us Measure 50 mL of Water (Page 144)
Place a clean, dry measuring cylinder on a flat surface.
Read the mark on the measuring cylinder that coincides with the bottom of the meniscus for water or other colourless liquids.
Answer:
While reading the measuring cylinder, the eye level should be on the lower meniscus for colourless liquid and on the top or upper meniscus for coloured liquid.